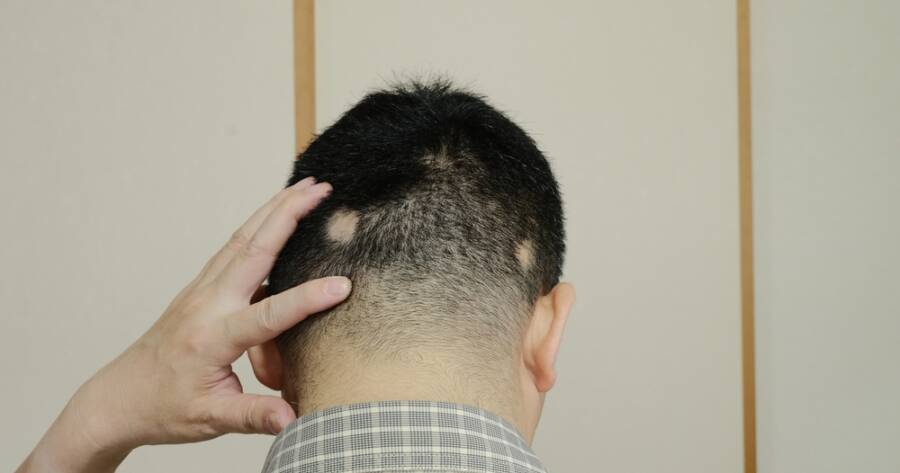
The FDA approval of LITFULO marks a transformative breakthrough in the treatment of alopecia areata, an autoimmune disorder marked by sudden hair loss affecting millions in the U.S. Groundbreaking medication offers new hope through its effective mechanism as a Janus kinase inhibitor, providing substantial hair regrowth and newfound confidence for patients, both adults and adolescents.
Understanding FDA-Approved Therapy for Alopecia Areata
Alopecia areata is an autoimmune disorder characterized by sudden and often unpredictable hair loss. Affecting approximately 7 million people across the United States, it can lead to considerable psychological and social challenges, particularly for young individuals. Until recently, effective treatments were limited, but the approval of LITFULO represents a groundbreaking development in this area.
As the first FDA-approved medication for treating severe alopecia in both adults and adolescents starting at age 12, LITFULO has shifted the paradigm of managing this condition by making strides in available therapies. Its approval offers new hope to those suffering from the physical and emotional burdens of the disorder.
The Mechanism Behind LITFULO
LITFULO works primarily as a Janus kinase (JAK) inhibitor, targeting JAK3 and TEC family kinases, which play critical roles in the autoimmune processes driving alopecia areata. By blocking specific cellular signals, it effectively tackles inflammation and contributes to significant hair regrowth.
In clinical trials, LITFULO demonstrated impressive efficacy, with results showing that 23% of participants achieved 80% or more scalp hair coverage after just six months of treatment, compared to a mere 1.6% with a placebo. This highlights its potential as a transformative treatment option for those who have previously had limited success with therapies like steroid injections.
Clinical Trial Success and Patient Impact
The impact of LITFULO and other JAK inhibitors, such as baricitinib, has been validated through numerous clinical trials. In particular, the ALLEGRO Phase 2b/3 trial shed light on LITFULO’s effectiveness by involving 718 patients who had experienced at least 50% scalp hair loss. The trial results were groundbreaking, revealing how LITFULO substantially helped participants regrow lost hair and regain confidence.
For many patients, the rapid progress made with LITFULO in as little as four months has been life-changing. Beyond the physical changes, the medication offers psychological relief from the embarrassment and anxiety associated with visible hair loss, particularly among adolescents.
Adverse Events and Considerations
As with any medication, patients using LITFULO may experience side effects. Common adverse events include headaches, diarrhea, acne, rashes, and urticaria, reported in at least 4% of trial participants.
It’s critical for those considering this treatment to discuss these potential adverse effects with their dermatologist to ensure an informed decision. Despite these potential side effects, the consistent safety profile observed across different age groups provides reassurance about the medication’s broader applicability for managing severe alopecia areata.
Financial Accessibility and Support
The availability of financial assistance programs aims to improve access to LITFULO, significantly enhancing patient adherence to the treatment regimen. Eligible patients can access the medication at reduced costs, sometimes paying as little as $0.
This financial support is crucial in ensuring that the benefits of LITFULO are accessible to a wider range of individuals suffering from alopecia areata, thereby extending the medication’s impact beyond just those who can afford it.
The Future of Alopecia Areata Treatment
While LITFULO and baricitinib represent significant advances in the treatment of severe alopecia areata, the work doesn’t stop here. The ongoing research into other potential JAK inhibitors such as deuruxolitinib aims to broaden the scope of effective therapies available to individuals affected by this condition.
Specialists emphasize the importance of ongoing innovation to develop solutions that cater to all patients, including those for whom current JAK inhibitors may not be effective. The goal is to ensure comprehensive coverage and treatment options for the various manifestations of alopecia areata.
Learn More About FDA-Approved Therapies for Alopecia Areata
The introduction of FDA-approved therapies like LITFULO marks a new era for alopecia areata management, offering solutions that are more effective and better targeted. Understanding these new options is crucial for both patients and healthcare providers aiming to improve the quality of life for those with this challenging condition.
Engaging with innovative treatments not only highlights the advances in medical science but also ensures that individuals affected by alopecia areata have access to the most current and effective interventions available. As research continues, keeping abreast of new developments is key to fostering hope and improving outcomes for millions affected by alopecia areata.
Sources
Yale News: Approval of Ritlecitinib
Yale Medicine: New Alopecia Treatment for Adolescents
Pfizer Press Release: Approval of LITFULO